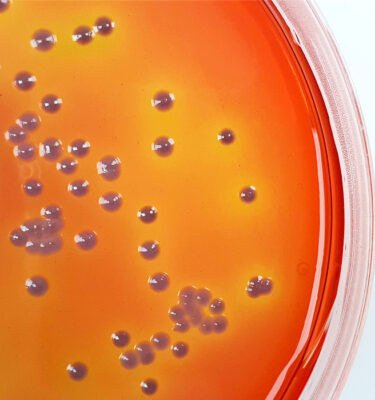
ABOUT SGL
Established over 95 years ago to manufacture and supply prepared culture media to the UK health service SOUTHERN GROUP LABORATORY (SGL) is now an independently owned media manufacturer that offers an extensive range of pre-poured bottled media, plated media, stains, reagents, and media for specialist applications across all sectors conducting Microbiology.
MAKING MEDIA FOR OVER 90 YEARS
Still a major supplier into clinical laboratories SGL now offers products into a diverse range of microbiology applications. Demand across all sectors for consistent, high quality prepared media has grown as companies look to enable more efficient workflow and minimise cost. SGL has developed in depth knowledge of each industry’s specific needs, knowledge of standards and regulatory requirements, production requirements and other industry specific concerns in order to deliver high quality products in line with customer expectation. High tech automated production in controlled environments ensures product meets the exacting standards required in modern microbiology.
Two enduring values underpin continued development
The Quality of our products & Passion for delivering the highest level of Service
FACILITIES
SGL supplies a wide range of industries including pharmaceutical, cosmetic, water, food and confectionery companies both across the UK and internationally. In our state-of-the-art manufacturing facility, we have invested in equipment and facilities to meet the exacting standards of quality and customer service being demanded by today’s microbiologists. Both bottled media and pre-poured plates are manufactured in clean room environments under precisely-controlled conditions to ensure optimal performance, quality and maximum shelf life for all our media products.
Factors that affect media stability during manufacture, such as exposure to heat are tightly controlled; F0 (total heat applied) is measured for all autoclaved products. All pre poured plates are poured and dried in a class 100 air flow with carefully controlled humidity to ensure that plates are dry and sterile. Pre poured plates and bottled media are stored under carefully controlled conditions prior to dispatch, ensuring that our products when they reach the customer are of the highest quality available.
- All activities are certified to ISO 9001 – Current edition, with quality control accredited by UKAS to EN ISO/ IEC 17025, ensuring consistent high quality products with continual improvement of processes and services in line with customer requirements
- All production takes place in a clean room environment. Our poured plate production facility has clean-rooms validated to: ISO 14644-1 (Class 7), EU GMP (Grade C) and all poured plate manufacturing is on fully automated production lines
- GMP and GLP techniques employed throughout all production and laboratory areas
- Rigorous QC examination with certificates available online
WHY USE SGL?
TIME
Extensive range of Prepared Culture Media available from stock next day.
CONSISTENCY
Highly consistent Prepared Culture Media every time.
QUALITY
Analysis carried out for you by our UKAS ISO: 17025 accredited testing laboratory
COST
Proven more cost effective than making Culture Media in house.
SERVICE
Standard formulation or Custom Prepared Media it’s your choice
QUALITY
QUALITY SYSTEM
Southern Group Laboratory products are manufactured at our Corby facility in compliance with our ISO 9001 Quality System. Every stage of manufacture is carefully controlled and documented to ensure the products are of consistently high quality. Representative samples of our products are QC tested in our on-site laboratory which is UKAS accredited to ISO 17025 and staffed by trained, experienced analysts.
The UKAS accredited QC tests are based on ISO 11133; methods have been validated and we have a programme of calibrations and operator checks to ensure the results are accurate.
Tests include the following:
All sterile media is tested for between 5 and 21 days at appropriate temperatures – usually 22 – 25oC, 35 – 37oC and 42 – 45oC to confirm sterility. In addition, we perform pH and gravimetric volume tests where these are relevant to the performance of the media.
Challenge testing of bottled and plated media to verify growth of target organisms or inhibition of non-target organisms meets pre-defined acceptance limits. This may be quantitative (where percentage recovery is specified) or qualitative.
Diluents and transport media are tested to ensure organism numbers remain constant over time and selective enrichment broths are tested to ensure they can recover the target organisms from mixed cultures of competing organisms.
Our product testing methods and specifications are derived from ISO 11133, Manufacturer’s recommendations or the relevant Pharmacopoeia to ensure each batch of media is fit for purpose. However, we can also customise testing to meet the needs of individual customers.
Certificates of analysis are designed for ease of use and to ensure all relevant information is available as quickly as possible. Certificates are available online.
If you have particular media or QC requirements, please contact us and set up your own formulation or testing specification.
Quality Downloads
ACCREDITATIONS
SGL are certified to the current version of ISO 9001 for the Development, Manufacture and Supply of Microbiological Culture Media. The Quality management system is designed and written to ensure accuracy and traceability throughout the business. “The development, manufacture and supply of microbiological culture media and associated equipment”
SGL’s QC Laboratory is accredited by UKAS in compliance with ISO/IEC 17025 for culture media test methods as per the accreditation schedule. Methodology is based on ISO 11133 and has been developed to ensure every batch of media is challenged both physically and microbiologically to ensure consistent quality throughout. You view the full schedule here.

SGL is a certified MHRA (Medicines & Healthcare products Regulatory Agency) supplier, offering a wide array of clinical reagents. Our products bear the CE mark, aligning with the European In-Vitro Diagnostics Medical Devices Directive 98/79/EC, ensuring quality and regulatory compliance in the field of healthcare and diagnostics.
FEATURED PRODUCTS
-
 Quick ViewApplication, Bacteria & Fungi, Bottle, Bottled and Bagged Media, By Product Group, By Sector, Clinical & Veterinary, Dairy, Environmental Monitoring, Featured, Food & Beverage, General Purpose, Needle-Free Media Bags, Organism, Pharma, Pharmaceutical & Cosmetic, Process Validation, Sterility Testing, Type, Water & Environmental
Quick ViewApplication, Bacteria & Fungi, Bottle, Bottled and Bagged Media, By Product Group, By Sector, Clinical & Veterinary, Dairy, Environmental Monitoring, Featured, Food & Beverage, General Purpose, Needle-Free Media Bags, Organism, Pharma, Pharmaceutical & Cosmetic, Process Validation, Sterility Testing, Type, Water & EnvironmentalTryptone Soya Broth, Bag with Spike Port & Capped Needle-Free Port
Read more -
 Quick ViewApplication, Bacteria & Fungi, Bottle, Bottled and Bagged Media, By Product Group, By Sector, Clinical & Veterinary, Dairy, Environmental Monitoring, Featured, Food & Beverage, General Purpose, Needle-Free Media Bags, Organism, Pharma, Pharmaceutical & Cosmetic, Process Validation, Sterility Testing, Type, Water & Environmental
Quick ViewApplication, Bacteria & Fungi, Bottle, Bottled and Bagged Media, By Product Group, By Sector, Clinical & Veterinary, Dairy, Environmental Monitoring, Featured, Food & Beverage, General Purpose, Needle-Free Media Bags, Organism, Pharma, Pharmaceutical & Cosmetic, Process Validation, Sterility Testing, Type, Water & EnvironmentalTryptone Soya Broth, Bag with Spike Port & Needle-Free Port
Read more -
Stericheck™ OVP- C
Read more -
Burkholderia Cepacia Selective Agar (BCSA), USP
Read more -
TSA Aircheck® 4W (Tryptone Soya Agar), 90mm Plate, Irradiated
Read more -
Stericheck™ OVP-B
Read more -
Stericheck™ OVP-A
Read more -
Quick ViewApplication, Bacteria & Fungi, Bio Burden, By Product Group, By Sector, Clinical & Veterinary, Contact Plate, Environmental Monitoring, Feature Page, Featured, General Purpose, Irradiated Plates, Organism, Pharma, Pharmaceutical & Cosmetic, Pre-Poured Plates, Surface Monitoring, Type, Water & Environmental
TSA Surfacecheck® (Tryptone Soya Agar), Contact Plate, Irradiated
Read more